
As Seen In: In Store September 2016
September 20, 2016
Fairy Tale Jewelry
October 21, 2016Our President & CEO, Kristie Nicolosi, traveled last month to Vincenzo for the Vincenza Oro jewelry show. Seeing so much beautiful gold jewelry at the show, she was inspired to reflect more on the different types of gold and its scientific properties. Gold has been considered a precious metal since ancient times, due to its luster, tarnish resistance, and the ease with which it can be worked.
Gold is used primarily for jewelry, though it has other uses in technology, manufacturing and even dental work. Gold itself is a softer metal, so it is alloyed with other metals, including nickel, copper, silver, zinc and palladium, to strengthen it.
Most of us are familiar with the measurement of alloys in gold, but might not realize it. Gold purity is measured using karats. One karat is equivalent to a 1/24 portion of pure gold. Thus 24 karat gold is pure gold, while 18 karat gold is 18 parts gold and 6 parts another metal. To be considered gold jewelry, a piece cannot be less than 10 karat.
Gold jewelry is available in a range of colors, and these hues are determined by the metals alloyed with the pure gold. Gold with higher percentages of copper alloys will take on a rose hue. Higher percentages of palladium, silver and nickel will produce white gold. Yellow golds are also an alloy but contain differing percentages of other metals.
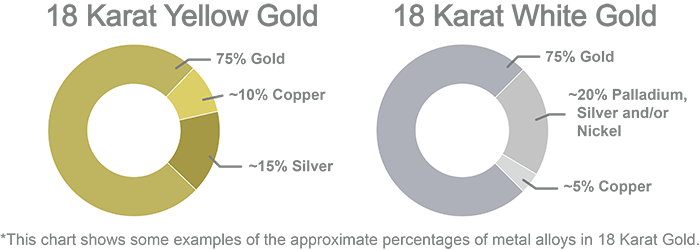
For example, an 18 karat yellow gold can be 75 percent gold, 15 percent silver and 10 percent copper, while an 18 karat white gold can be 75 percent gold, 5 percent copper and 20 percent palladium, silver and/or nickel. Both shades of gold are 18 karat and therefore contain 75 percent gold, but the other metals in the alloy are what determine its hue. Some jewelry designers only use a certain shade of gold in their work, and thus have a specific alloy formula which they purchase for their designs.
The chemical composition of gold has effects other than just the shade of gold produced. The metals in a piece of gold can react with chemicals in cleaning agents, cosmetics and the like, so it is recommended to remove jewelry when it will come in contact with these substances. While the body chemistry of some individuals can also react with the metal alloys causing skin to turn black or green or even discolor the jewelry. This can be accentuated due to conditions such as anemia (an iron deficiency) or due to medications, particularly antibiotics. The higher the karat of the jewelry, the less likely there will be a reaction, as there is a lower percentage of alloyed metals.
When cleaning gold jewelry, it’s important to be aware that the gold does contain other metals. Fine jewelry cleaning formulas are safe for most pieces, though if your jeweler is unsure of the composition of the piece, using a gentle jewelry cleaning formula may be a better choice. Be sure to follow the care instructions carefully, particularly with regard to the length of time the piece is submerged. If there is any tarnish on a piece (gold cannot tarnish, but some alloyed metals can), a professional polishing cloth can remove tarnish or discoloration.






